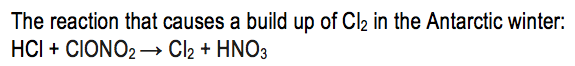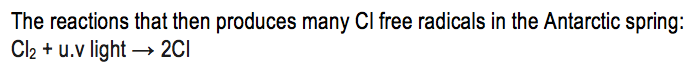Origins of CFC's
Haloalkanes are compounds in which one or more hydrogen atom(s) of an alkane has been replaced by a halogen atom. Chlorofluorocarbons or CFC’s are haloalkane molecules in which all the hydrogen atoms have been replaced by fluorine or chlorine atoms. And example is CFC-11, also known by its systematic name as trichlorofluromethane, which was used as fluid in refrigeration and air conditioning and has the structural formula CCl3F. Halons are haloalkane molecules in which all the hydrogen atoms have been replaced by bromine, chlorine and/or fluorine. CFC’s were introduced in the 1930’s to replace toxic ammonia gas in refrigeration because they were unreactive, non-toxic and readily liquefied upon compression, which made them ideal working fluids. Three other applications of CFC’s include being used as propellants in spray cans, as ‘blowing’ agents to make expanded plastic and as solvents for dry cleaning and cleaning electrical circuits. A past use of halons was as fire extinguishers, as they are dense non-flammable liquids.
CFC’s have the unique properties of being odourless, non-flammable, non-toxic, insoluble in water and very inert, and because of these properties initially CFC’s were thought to be of no concern and so they were freely released into the atmosphere. However because CFC’s are inert and insoluble in water (rain) they began to become a problem, as they were not being destroyed in the atmosphere by sunlight or oxygen like most organic compounds. The CFC’s spread out around the world, and their concentrations in the troposphere gradually built up until they diffused into the stratosphere where they began to cause a problem. In the stratosphere CFC’s come into contact with short wavelength u.v radiation and this breaks a chlorine atom off the CFC molecule, which then abstracts an oxygen atom from an ozone molecule, forming a ClO free radical, which is very reactive and continues to form with and destroy many ozone molecules.
CFC’s have the unique properties of being odourless, non-flammable, non-toxic, insoluble in water and very inert, and because of these properties initially CFC’s were thought to be of no concern and so they were freely released into the atmosphere. However because CFC’s are inert and insoluble in water (rain) they began to become a problem, as they were not being destroyed in the atmosphere by sunlight or oxygen like most organic compounds. The CFC’s spread out around the world, and their concentrations in the troposphere gradually built up until they diffused into the stratosphere where they began to cause a problem. In the stratosphere CFC’s come into contact with short wavelength u.v radiation and this breaks a chlorine atom off the CFC molecule, which then abstracts an oxygen atom from an ozone molecule, forming a ClO free radical, which is very reactive and continues to form with and destroy many ozone molecules.
Problems Cause by CFC's
The ozone hole is the thinning of the ozone layer. In the 1980’s the ozone lose worsened and the area it spread over became wider. The depletion of ozone and the worsening ozone hole above Antarctica were first mentioned in a paper in 1985. Ozone depletion is not limited to the Antarctic, as it has been recorded that during early summer ozone depleted stratospheric air has spread over Tasmania and Victoria before. The ozone hole is the thinning of the ozone above the Antarctic, where as ozone depletion is the decrease in the amount of ozone in the stratosphere. u.v-a has a wavelength range of 400 to 320 nm where as u.v-b has a wavelength range of 320-280 nm. u.v-a is generally considered beneficial because it facilitates photosynthesis and helps form Vitamin D in humans and it also produces a suntan, where as u.v-b is considered harmful because it causes skin cancer, eye cataracts, decreased immune response, it damages plants and also causes severe sunburn. Ozone depletion leads to more u.v-b reaching earths surface because short wavelength u.v-b acts upon oxygen gas to form oxygen atoms which then combine with O2 molecules in the air to form ozone and this ozone in turn absorbs longer wavelength u.v which decomposes the ozone back to ordinary oxygen, thus the longer wavelength u.v-a is absorbed while the short wavelength u.v-b destroys the ozone and reaches earth. Stratospheric ozone depletion leads to more u.v-b reaching the earths surface which in humans can cause skin cancer, eye cataracts, decreased immune response and severe sunburn as well as interfere with the mechanisms for photosynthesis in some plants.
In the stratosphere CFC’s come into contact with short wavelength u.v which breaks a chlorine atom off the CFC molecule, CCl2F2 + u.v light → Cl* + CCIF2. The chlorine atom then extracts an oxygen atom from an ozone molecule, forming a ClO free radical, Cl* + O3 → ClO* + O2. The ClO, like all free radicals, is very reactive and so reacts with a free oxygen, present from the continual formation and destruction of ozone in the stratosphere, to form a molecule of oxygen (O2) and this regenerates the chlorine atom, ClO* + O* → O2 + Cl*. The result of these equations is that an ozone molecule and an oxygen atom have been converted into two oxygen molecules and our reactive chlorine atom has not been used up. Therefore this chlorine atom is able to attack other ozone molecule and repeat the process over again and again. Therefore the chlorine free radicals are catalysts for ozone destruction and hence one CFC molecule can do tremendous damage.
This chain reaction of Cl free radicals can be brought to a halt by a reaction between a chlorine atom and a methane molecule, which forms hydrogen chloride: Cl* +CH4 → HCl + CH3*. This reaction removes a chlorine atom and so brings the chain reaction to a halt, as HCl has effect upon ozone and so reactive Cl atoms are converted into unreactive HCl molecules. It can also be brought to a halt by a reaction involving the ClO species: ClO* + NO2 → ClONO2. Small amounts of NO2 are present in the stratosphere and the occurrence of this reaction essentially removes ClO from the above chain reaction and brings the destruction of ozone to a halt, especially as Cl atoms cannot be regenerated from ClONO2. Before this halt in the chain reaction occurs one CFC molecule can destroy thousands or perhaps tens of thousands of ozone molecules, which is why the very small concentrations of CFC’s that actually diffuse into the stratosphere can cause significant damage.
Two feature of the Antarctic winter that help to reduce ozone depletion are that the winter is a period of continues darkness and that the stratosphere there is extremely cold due to the polar vortex. Under these extremely cold conditions certain solid particles form that are able to catalyse a reaction between hydrogen chloride and chlorine nitrate: HCl + ClONO2→ Cl2 + HNO3. This conversion of hydrogen chloride and chlorine nitrate to molecular chlorine has no effect upon the ozone concentrations during winter and does not deplete it. However in the spring, when the sun comes up, sunlight is able to split a chlorine molecule into two separate chlorine atoms: Cl2 + u.v light → 2Cl. Each of these chlorine atoms can destroy ozone in exactly the same way as chlorine atoms formed from CFC’s. Therefore in spring there is an extra source of chlorine atoms and so consequently increased destruction of ozone and a dramatic decrease in ozone levels.
This rate of ozone destruction however doesn’t continue into the summer because during the polar winter only a finite amount of molecular chlorine is formed and by early summer this has been virtually all used up, so the rate of destruction of ozone returns to the ‘normal’ CFC level. Also the polar vortex breaks up in early summer and so and so there is a mixing of Antarctic air with lower-altitude air, which brings the ozone concentration back to its normal level.
The Antarctic ozone hole poses problems for countries in the southern hemisphere because the break up of the polar vortex has occasionally brought ozone depleted stratospheric air over Tasmania, parts of Victoria and New Zealand in early summer. This is a problem as it allows more u.v radiation to reach ground level, which can lead to increase amounts of people contracting skin cancer and the death of vital surface dwelling phytoplankton and zooplankton, which would produce a significant decline in marine organisms.
In the stratosphere CFC’s come into contact with short wavelength u.v which breaks a chlorine atom off the CFC molecule, CCl2F2 + u.v light → Cl* + CCIF2. The chlorine atom then extracts an oxygen atom from an ozone molecule, forming a ClO free radical, Cl* + O3 → ClO* + O2. The ClO, like all free radicals, is very reactive and so reacts with a free oxygen, present from the continual formation and destruction of ozone in the stratosphere, to form a molecule of oxygen (O2) and this regenerates the chlorine atom, ClO* + O* → O2 + Cl*. The result of these equations is that an ozone molecule and an oxygen atom have been converted into two oxygen molecules and our reactive chlorine atom has not been used up. Therefore this chlorine atom is able to attack other ozone molecule and repeat the process over again and again. Therefore the chlorine free radicals are catalysts for ozone destruction and hence one CFC molecule can do tremendous damage.
This chain reaction of Cl free radicals can be brought to a halt by a reaction between a chlorine atom and a methane molecule, which forms hydrogen chloride: Cl* +CH4 → HCl + CH3*. This reaction removes a chlorine atom and so brings the chain reaction to a halt, as HCl has effect upon ozone and so reactive Cl atoms are converted into unreactive HCl molecules. It can also be brought to a halt by a reaction involving the ClO species: ClO* + NO2 → ClONO2. Small amounts of NO2 are present in the stratosphere and the occurrence of this reaction essentially removes ClO from the above chain reaction and brings the destruction of ozone to a halt, especially as Cl atoms cannot be regenerated from ClONO2. Before this halt in the chain reaction occurs one CFC molecule can destroy thousands or perhaps tens of thousands of ozone molecules, which is why the very small concentrations of CFC’s that actually diffuse into the stratosphere can cause significant damage.
Two feature of the Antarctic winter that help to reduce ozone depletion are that the winter is a period of continues darkness and that the stratosphere there is extremely cold due to the polar vortex. Under these extremely cold conditions certain solid particles form that are able to catalyse a reaction between hydrogen chloride and chlorine nitrate: HCl + ClONO2→ Cl2 + HNO3. This conversion of hydrogen chloride and chlorine nitrate to molecular chlorine has no effect upon the ozone concentrations during winter and does not deplete it. However in the spring, when the sun comes up, sunlight is able to split a chlorine molecule into two separate chlorine atoms: Cl2 + u.v light → 2Cl. Each of these chlorine atoms can destroy ozone in exactly the same way as chlorine atoms formed from CFC’s. Therefore in spring there is an extra source of chlorine atoms and so consequently increased destruction of ozone and a dramatic decrease in ozone levels.
This rate of ozone destruction however doesn’t continue into the summer because during the polar winter only a finite amount of molecular chlorine is formed and by early summer this has been virtually all used up, so the rate of destruction of ozone returns to the ‘normal’ CFC level. Also the polar vortex breaks up in early summer and so and so there is a mixing of Antarctic air with lower-altitude air, which brings the ozone concentration back to its normal level.
The Antarctic ozone hole poses problems for countries in the southern hemisphere because the break up of the polar vortex has occasionally brought ozone depleted stratospheric air over Tasmania, parts of Victoria and New Zealand in early summer. This is a problem as it allows more u.v radiation to reach ground level, which can lead to increase amounts of people contracting skin cancer and the death of vital surface dwelling phytoplankton and zooplankton, which would produce a significant decline in marine organisms.
Reactions in the Atmosphere
Monitoring the Ozone Layer
The
Australian Bureau of Meteorology monitors the ozone in the atmosphere at
different altitudes using balloons and aircraft. They also use a ground-based Dobson
spectrometer to monitor stratospheric ozone. Since the 1970’s NASA has been
monitoring the concentration of stratospheric ozone via a spectrometer in a
satellite. NASA’s total ozone mapping spectrometer (TOMS) measures certain
frequencies of sunlight scattered back into space by the earth’s atmosphere. In
2012 this was was replaced by The Ozone Mapper and profiler Suite (OMPS).
An advantage of using satellites such as TOMS and OMPS to measure stratospheric ozone is that it provides relatively accurate maps showing the ozone concentration from ground level to space over most of the earth, including the atmosphere over the oceans where ground instruments are not possible. Another advantage is that these satellites produce more accurate data than most other satellites. A disadvantage of this technique is that data from satellites such as TOMS and OMPS is not available in real time and is received routinely via the World Meteorological Organisation's Global Telecommunications System.
An advantage of using ground-based equipment such as the Dobson Spectrophotometer is that it can very accurately estimate total column ozone amounts and produce reliable data. A disadvantage with this technique is that the information is not readily available as the whole process, from the time measurements are taken until they are properly quality controlled, can take 3 to 4 months.
An advantage of using balloons or Ozone sonde’s is that they are the most accurate means of determining at what heights ozone variations are occurring and that the data collected from them greatly assists researchers in pinpointing whether the variations are natural or anthropogenic in origin. A disadvantage is that the utmost care in cleanliness, skill in calibration and procedures must be used when handling the machinery, which can be time consuming and difficult. Another disadvantage is that the readings taken by the Ozone Sonde are affected if pollutants, especially sulfur dioxide and nitrous oxide, are present in the atmosphere as these can interfere with the measurement of ozone. Another disadvantage is that the balloon has a burst point, typically in excess of 30 km, which means that past this point the Ozone Sonde cannot continue measuring.
A Dobson unit (DU) is the amount of ozone that corresponds to a 0.01mm thickness of pure ozone at atmospheric pressure. The normal amount of ozone is 300 DU.
An advantage of using satellites such as TOMS and OMPS to measure stratospheric ozone is that it provides relatively accurate maps showing the ozone concentration from ground level to space over most of the earth, including the atmosphere over the oceans where ground instruments are not possible. Another advantage is that these satellites produce more accurate data than most other satellites. A disadvantage of this technique is that data from satellites such as TOMS and OMPS is not available in real time and is received routinely via the World Meteorological Organisation's Global Telecommunications System.
An advantage of using ground-based equipment such as the Dobson Spectrophotometer is that it can very accurately estimate total column ozone amounts and produce reliable data. A disadvantage with this technique is that the information is not readily available as the whole process, from the time measurements are taken until they are properly quality controlled, can take 3 to 4 months.
An advantage of using balloons or Ozone sonde’s is that they are the most accurate means of determining at what heights ozone variations are occurring and that the data collected from them greatly assists researchers in pinpointing whether the variations are natural or anthropogenic in origin. A disadvantage is that the utmost care in cleanliness, skill in calibration and procedures must be used when handling the machinery, which can be time consuming and difficult. Another disadvantage is that the readings taken by the Ozone Sonde are affected if pollutants, especially sulfur dioxide and nitrous oxide, are present in the atmosphere as these can interfere with the measurement of ozone. Another disadvantage is that the balloon has a burst point, typically in excess of 30 km, which means that past this point the Ozone Sonde cannot continue measuring.
A Dobson unit (DU) is the amount of ozone that corresponds to a 0.01mm thickness of pure ozone at atmospheric pressure. The normal amount of ozone is 300 DU.
The Montreal Protocol & Alternatives to CFC's
HCFC’s are hydrochlorofluorocarbons,
compounds containing C-H bonds, which are susceptible to attack by reactive
radicals and atoms in the troposphere and so are decomposed to a significant
extent. Consequently there ozone depletion potential is much less than CFC’s
but is still significant. HCFC’s are considered a temporary replacement for CFC’s
because while being less destructive to the ozone than CFC’s they are
greenhouse gases and are quite slow to be destroyed, which means substantial
portions of intact molecules still reach the stratosphere where they have ozone
depletion potential. They’re also
considered temporary because they contain chlorine atoms and thus C-Cl bonds,
so when they are released into the stratosphere the u.v radiation breaks the
bond and creates Cl free radicals, which will destroy between 1-10% of as much
ozone as the same amount of CFC’s, which is significantly less than CFC’s but
still quite destructive. Other disadvantages of HCFC’s include that some types
can be quite corrosive or flammable while other HCFC’s without these properties
tend to be quite expensive. The substitution of HCFC’s also requires expensive
system redesigns of appliances such as refrigerators and air conditioners.
HCFC’s also have a high global warming potential and are significantly more
damaging than CO2 (the standard).
HFC’s are hydrofluorocarbons, compounds containing hydrogen, fluorine and carbon but no chlorine. They contain C-H bonds so they undergo some decomposition in the troposphere but contain no C-Cl bonds, unlike CFC’s or HCFC’s, so they do not form Cl atoms in the stratosphere and therefore have zero ozone depletion potential which is why they are considered a better replacement than HCFC’s. Some disadvantages of HFC’s include that they are greenhouse gases which means that they contribute significantly to global warming and have a high global warming potential, they can be quite corrosive, flammable and expensive and they require expensive system redesign when being implemented.
HCFC’s and HFC’s cannot replace all CFC’s as the do not have all the same properties and capabilities as some CFC’s, therefore they cannot properly fill the role of some CFC’s. It’s also very costly to replace all CFC’s and many third world countries that use CFC’s do not have the money or resources to implement fazing out of CFC’s. Several HCFC and HFC alternative also have high global warming potential, over 1000 time that of CO2, which makes them more toxic, corrosive, explosive or flammable than the CFC’s they are replacing.
HFC’s are hydrofluorocarbons, compounds containing hydrogen, fluorine and carbon but no chlorine. They contain C-H bonds so they undergo some decomposition in the troposphere but contain no C-Cl bonds, unlike CFC’s or HCFC’s, so they do not form Cl atoms in the stratosphere and therefore have zero ozone depletion potential which is why they are considered a better replacement than HCFC’s. Some disadvantages of HFC’s include that they are greenhouse gases which means that they contribute significantly to global warming and have a high global warming potential, they can be quite corrosive, flammable and expensive and they require expensive system redesign when being implemented.
HCFC’s and HFC’s cannot replace all CFC’s as the do not have all the same properties and capabilities as some CFC’s, therefore they cannot properly fill the role of some CFC’s. It’s also very costly to replace all CFC’s and many third world countries that use CFC’s do not have the money or resources to implement fazing out of CFC’s. Several HCFC and HFC alternative also have high global warming potential, over 1000 time that of CO2, which makes them more toxic, corrosive, explosive or flammable than the CFC’s they are replacing.
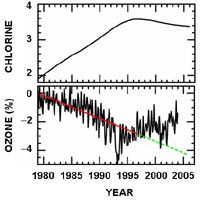 Graph 1: NASA/NOAA satellite data showing the rise in stratospheric chlorine and corresponding decline in ozone layer thickness from 1979 to 1997
Graph 1: NASA/NOAA satellite data showing the rise in stratospheric chlorine and corresponding decline in ozone layer thickness from 1979 to 1997
Since HCFC’s and HFC’s have been
introduced, which occurred in 1989, there has been a rise in stratospheric
chlorine between 1989-1995, then a levelling out between period in between and
a slight decline in the amount of stratospheric chlorine between 1998-2005.
Correspondingly there was a dramatic drop in the percentage thickness of the
ozone layer between 1989-1995 but then an overall increase in ozone thickness
between 1995-2005.
The
effectiveness of HFC’s and HCFC’s can be judged by how well they replace CFC’s,
their effect on the ozone and their contribution to the greenhouse effect. HFC’s
and HCFC’s have not yet been developed so that they can effectively replace all
the functions of CFC’s. Many suggested and implement HFC and HCFC replacements
are highly toxic, flammable, corrosive and expensive which does not make them
effective replacements for CFC’s. HCFC’s contain chlorine atoms and so still
cause some destruction of the ozone but far less than CFC’s, and HFC’s cause no
harm to ozone as they contain no chlorine atoms and have zero ozone depletion
potential, which makes them effective replacements for CFC’s. . HFC’s and
HCFC’s are greenhouse gases and so when released into the atmosphere the absorb
heat and contribute to global warming. HFC’s and HCFC’s have global warming potential, over 1000 time that of CO2, which
doesn’t makes them effective replacements for CFC’s. I don’t think HFC’s and HCFC’s
are an effective replacement for CFC’s because they are not able to fully
replace the operations of many CFC’s, are very expensive and can be flammable,
toxic and corrosive and are greenhouse gases with very high global warming
potential.
The purpose of the Montreal protocol is to be an international treaty in which countries agree to create a time plan to phase out the production of numerous substances that are responsible for ozone depletion such as CFC’s. The treaty came into effect in 1989 and was revised in 1992, with many set dates as to when certain substances had to be phased out by or the use and production reduced to below a certain percentage. The use of halons was to be ceased by 1994. The manufacturing and use of CFC’s and 1,1,1-trichloroethane was to end by 1996. The phasing out of less active HCFC’s only began in 1996 and will continue until a complete phasing out by 2030. The phasing out of more active HCFC’s consumption and production was set for 2013 but was revised and set for 2015.
The purpose of the Montreal protocol is to be an international treaty in which countries agree to create a time plan to phase out the production of numerous substances that are responsible for ozone depletion such as CFC’s. The treaty came into effect in 1989 and was revised in 1992, with many set dates as to when certain substances had to be phased out by or the use and production reduced to below a certain percentage. The use of halons was to be ceased by 1994. The manufacturing and use of CFC’s and 1,1,1-trichloroethane was to end by 1996. The phasing out of less active HCFC’s only began in 1996 and will continue until a complete phasing out by 2030. The phasing out of more active HCFC’s consumption and production was set for 2013 but was revised and set for 2015.
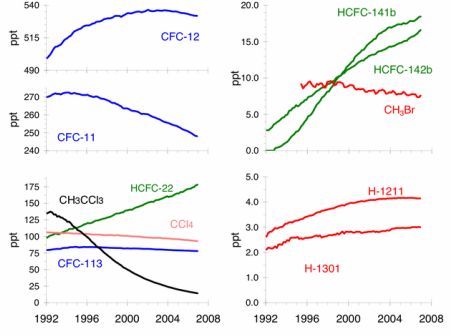 Graph 2: Ozone Depleting Gas trends
Graph 2: Ozone Depleting Gas trends
The Montreal protocol has been an
effective agreement as most countries have been able to meet the required
targets, due to the availability of HFC’s, which are acceptable alternative
compounds to CFC’s. Atmospheric
concentrations of CFC’s have either levelled or decreased but halons have
continued to increase, as they are used in fire extinguishers, but there rate
of increase has slowed and their abundances are expected to begin to decline by
2020. Unfortunately there has been a dramatic increase in the concentration of
HCFC’s as they are being used as substitution for CFC’s. Statistical analysis conducted
in 2010 showed a clear positive signal from the Montreal Protocol to the
stratospheric ozone. However HCFC’s are now know to contribute to anthropogenic
global warming and to have a global warming potential 1000 times more than CO2.
Consequently the complete phase out of HCFC’s has been set for 2030. Overall
the Montreal
protocol has been effective in implementing its phase out strategies and
is known as the most successful international environmental agreement to date.
is known as the most successful international environmental agreement to date.
The replacement
chemicals for CFC’s are HCFC’s. The main advantage of HCFC’s is that their ozone depletion potential is much less than CFC’s,
they only destroy between
1-10% of as much ozone as the same amount of CFC’s.
One of the main disadvantages
of HCFC’s is that they have a
high global warming potential and are up to more 1000 times more damaging than
CO2 (the standard). The other
main disadvantage is that they contain chlorine atoms and thus C-Cl bonds, so
when they are released into the stratosphere the u.v radiation breaks the bond
and creates Cl free radicals, which is quite destructive to ozone. Other
disadvantages of HCFC’s are that they can be corrosive, flammable, toxic and
expensive and require expensive system redesigns of appliances such as
refrigerators and air conditioners when being implemented to replace CFC’s.
CFC’s will persists in the atmosphere for some time because they are a greenhouse gas and are quite slow to be destroyed, which means substantial portions of intact molecules still reach the stratosphere where they have ozone depletion potential.
Since the implementation of the Montreal protocol there has not been a huge measured decrease in atmospheric concentrations of CFC’s but they have either leveled or decreased slightly. However the ozone hole has not been getting any shallower but significant progress has been made towards solving the ozone hole problem.
CFC’s will persists in the atmosphere for some time because they are a greenhouse gas and are quite slow to be destroyed, which means substantial portions of intact molecules still reach the stratosphere where they have ozone depletion potential.
Since the implementation of the Montreal protocol there has not been a huge measured decrease in atmospheric concentrations of CFC’s but they have either leveled or decreased slightly. However the ozone hole has not been getting any shallower but significant progress has been made towards solving the ozone hole problem.