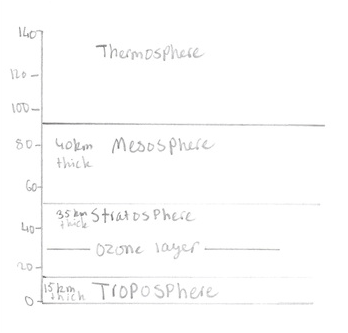
The Structure and Composition of the Atmosphere
Main gases of the troposphere
Nitrogen 78%
Oxygen 21%
Argon 0.9%
Carbon Dioxide 350ppm
Nitrogen 78%
Oxygen 21%
Argon 0.9%
Carbon Dioxide 350ppm
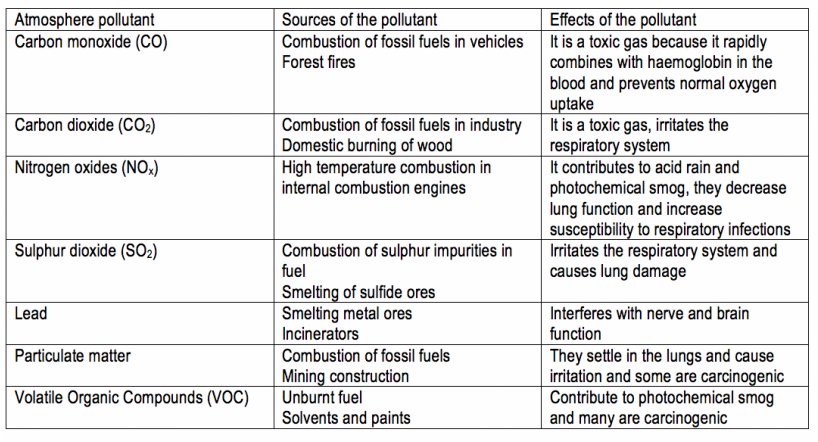
Atmospheric Pollutants
Ozone- Good and Bad?
The atmosphere contains less than 2ppb (by volume) of ozone. This is a small amount compared to nitrogen, 8.8 x108ppb and oxygen, 2.0 x108 ppb. About 10% of ozone is present in the troposphere. Smog is a type of air pollutant that is produced when sunlight acts upon motor car exhaust gases to form ozone and other harmful substances.
Ground level ozone forms when sunlight is very intense and when concentrations of NO2 are well above clear-air levels. This happens when there is no breeze to disperse pollutants. The sunlight splits off an oxygen atom from the NO2 molecule and this oxygen atom combines with an oxygen molecule, O2 to form ozone. NO2 + sunlight → NO + O, O + O2→ O3. Ground level ozone can be very poisonous. It irritates the eyes and causes breathing difficulties. It’s also toxic to plants, including agricultural crops. It is a strong oxidising agent and readily attacks rubber and plastics. To reduce ground level ozone the amounts of the pollutants nitrogen dioxide and nitrogen oxide being produced need to be reduce . Car exhaust is a major source of these pollutants and so to reduce the amounts of nitrogen dioxide and nitrogen oxide being produced cleaner alternatives need to be found such as electric cars.
In the stratosphere short wavelength u.v. acts upon oxygen gas to form oxygen atoms which then combine with O2 molecules to form ozone:
O2 + u.v. light → 2O, O + O2 → O3. Oxygen atoms can slowly react with ozone: O + O3 → 2O2. The ozone in turn absorbs longer wavelength u.v. which decomposes the ozone back to ordinary oxygen: O3 + u.v. light → O + O2. There is a balance between the formation and destruction of ozone in these four reactions, which leaves a ‘steady state’ concentration of ozone in the stratosphere. This stratospheric ozone is helpful to biological processes on the Earth’s surface because it acts as a shield against the u.v. radiation reaching the earths surface. This u.v. can be harmful to humans and cause sunburn and skin cancer and can also interfere with the mechanisms for photosynthesis in some plants.
Ground level ozone forms when sunlight is very intense and when concentrations of NO2 are well above clear-air levels. This happens when there is no breeze to disperse pollutants. The sunlight splits off an oxygen atom from the NO2 molecule and this oxygen atom combines with an oxygen molecule, O2 to form ozone. NO2 + sunlight → NO + O, O + O2→ O3. Ground level ozone can be very poisonous. It irritates the eyes and causes breathing difficulties. It’s also toxic to plants, including agricultural crops. It is a strong oxidising agent and readily attacks rubber and plastics. To reduce ground level ozone the amounts of the pollutants nitrogen dioxide and nitrogen oxide being produced need to be reduce . Car exhaust is a major source of these pollutants and so to reduce the amounts of nitrogen dioxide and nitrogen oxide being produced cleaner alternatives need to be found such as electric cars.
In the stratosphere short wavelength u.v. acts upon oxygen gas to form oxygen atoms which then combine with O2 molecules to form ozone:
O2 + u.v. light → 2O, O + O2 → O3. Oxygen atoms can slowly react with ozone: O + O3 → 2O2. The ozone in turn absorbs longer wavelength u.v. which decomposes the ozone back to ordinary oxygen: O3 + u.v. light → O + O2. There is a balance between the formation and destruction of ozone in these four reactions, which leaves a ‘steady state’ concentration of ozone in the stratosphere. This stratospheric ozone is helpful to biological processes on the Earth’s surface because it acts as a shield against the u.v. radiation reaching the earths surface. This u.v. can be harmful to humans and cause sunburn and skin cancer and can also interfere with the mechanisms for photosynthesis in some plants.